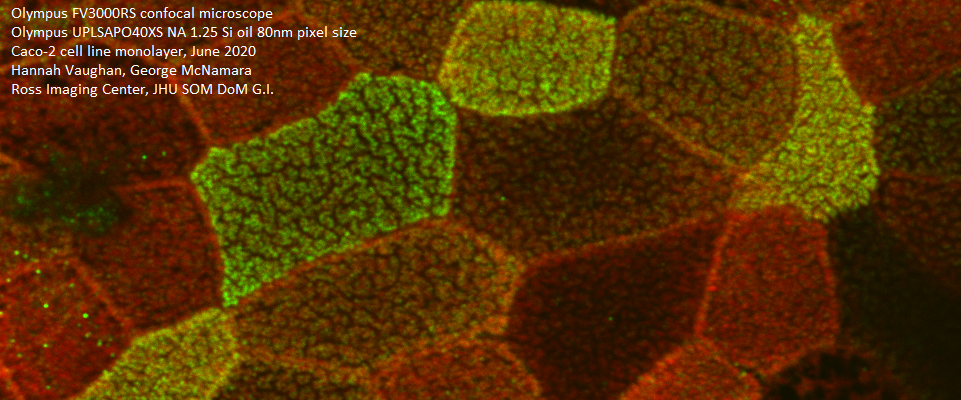
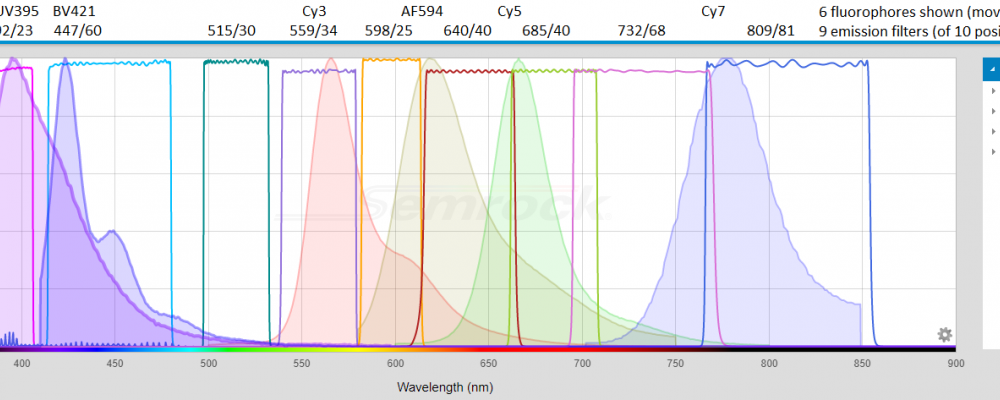
WELCOME
Ross Imaging Center is the image core for the Hopkins Conte Digestive Diseases Basic and Translational Research Core Center (http://jhugicc.org) is an NIH/NIDDK funded, institutionally supported center that has as its mission, to make gastrointestinal studies at Hopkins as outstanding as possible, by encouraging helping develop interactions between Hopkins basic and translational investigators. P30 Center grant P.I. Prof. Mark Donowitz.
Reservations (iLab URLs changed in 2024)
Ross image core https://johnshopkins.ilab.agilent.com/sc/5148/ross-imaging-center/?tab=equipment
ACCM LeicaSP8 confocal microscope https://johnshopkins.ilab.agilent.com/schedules/345585#/schedule
Ross Imaging Center = Core B: Imaging (http://jhugicc.org/cores/core-b-imaging)
Mission
Ross Fluorescence Imaging Center core is dedicated to providing state-of-art light microscopy technology to the members of the Hopkins Basic Research Digestive Disease Development Center and to the general Hopkins scientific community. The image core is open access to all at JHU (need iLab account or arrangement to pay by invoice), JHU FastForward incubator tenants, and non-JHU users, both academic (ex. UMd, other colleges & universities) and commercial (different rates may apply).
Image core manager George McNamara, PhD.
* as of 7/2023 George is 20% time in Ross bldg, 80% time in Smith Bldg managing the High Throughput Phenotypic Screening Core (HPS core) - please plan ahead for anything to do with the Ross Imaging Core or ACCM Leica SP8 confocal microscope core.
**
August 2024 iLab URL change - my JH link should update but you may need to manually fix your bookmarks:
https://johnshopkins.ilab-int.agilent.com
Full notice: Please be aware that as of 8/3/2024 (late Friday night) the URL for the Johns Hopkins University iLab website will be changing to https://johnshopkins.ilab-int.agilent.com. Any previously saved bookmarks for iLab should be updated accordingly. If you encounter any issues with logging into iLab with your JHED ID, please call the Johns Hopkins IT Help Desk at 410-516-HELP (410-516-4357) or submit a Johns Hopkins Help Desk self-service ticket online by accessing https://myit.jh.edu/apps/mfahelpticket.
**
Covid-19 pandemic precautions
August 1,2024: covid-19 is still around, and current variants more-or-less escape from immunity and are infecting people. If you are sick, please stay home. Historically, ~40% of infected adults are "non-symptomatic" -- but still can infect -- and some infected may be "super-spreaders" -- so if you may have been exposed, please stay home (the home test kits probably still detect current variants - but CDC etc have not made this clear and some new variant could appear that the current tests miss ... bonus: old test kits may not work in 2024).
September 14, 2022: JHU SOM In nonclinical buildings: As of Thursday, Sept. 15, masks are recommended but no longer required for JHM personnel working in nonclinical buildings where there is NO reasonable expectation of an encounter with a patient.
Ross Image Core policy - Sept 14, 2022: mask is optional for FULLY VACCINATED users -- as up to date covid=19 vaccines, boosters, new version(s) -- original+Onicron bivalent mRNA vaccines just came out. If you are not fully vaccinated, please wear a mask in (and near) the image core -- or arrange for a fully vaccinated colleague to do your imaging. If you do wear a mask, please wear it correctly.
Please continue to social distance as much as practical, that is one user per microscope station preferred, two ok, more is probably a bad idea.
old info - Covid-19 news Thursday July 29, 2021 (from today's JHMI email): (As of) Sept. 1, 2021 All Johns Hopkins Medicine personnel must be fully vaccinated* or receive an approved exception. Personnel should have received the first dose of the Moderna or Pfizer COVID-19 vaccine by mid-July, and should receive the second dose, or a single dose of the Johnson & Johnson vaccine, by mid-August to meet this deadline. Please remember that this requirement applies to all school of medicine faculty members, residents, clinical fellows, postdoctoral research fellows, medical students, graduate students and staff members. ... GM note: same wrt vendors and other visitors to research spaces.
even older info - Coronavirus news: Monday June 15, 2020 we re-opened as part of JHU-Research's covid-19 phase 1 re-opening. All users must read and follow our policies:
http://confocal.jhu.edu/covid-19-policies
***
June 22, 2020 Pricing information is at http://confocal.jhu.edu/facility-usage-fees
More news (and some tips) at http://confocal.jhu.edu/image-core-news
***
All reservations through iLab
ACCM Leica SP8 confocal microscope iLab
Ross Imaging Center iLab (information at top, links to instruments below)
More details at Current Equipment page.